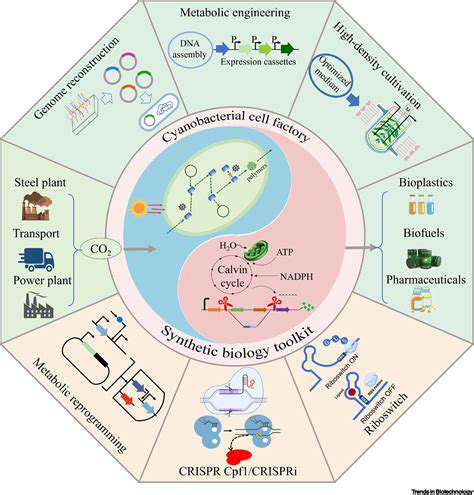
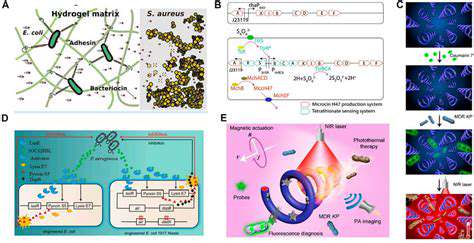
Microbial biofuel production represents a promising avenue for sustainable energy generation. Harnessing the metabolic capabilities of microorganisms offers a potentially renewable and environmentally friendly alternative to fossil fuels. This approach leverages the natural processes of microorganisms to convert various feedstocks into biofuels, minimizing the need for large-scale chemical processing. The efficiency and scalability of microbial biofuel production are key considerations for future development. This innovative technology holds the potential to address the growing global energy demand while mitigating the environmental impact of conventional energy sources.
Several microorganisms, including bacteria and yeasts, possess unique metabolic pathways that can be exploited for biofuel synthesis. These microorganisms are capable of converting organic matter into biofuels like ethanol, butanol, and hydrogen. The selection of appropriate microorganisms and optimal growth conditions are crucial factors in maximizing biofuel yield and minimizing production costs. Furthermore, the choice of feedstock significantly impacts the overall sustainability and economic viability of the process.
Key Considerations in Microbial Biofuel Production
One critical aspect of microbial biofuel production is the selection of appropriate feedstocks. The feedstock should be readily available, cost-effective, and sustainable to minimize the environmental footprint. Agricultural residues, industrial waste, and even municipal solid waste can serve as potential feedstocks. Careful consideration of the feedstock's composition and characteristics is necessary to optimize the microbial conversion process.
Another important factor is the optimization of the microbial culture conditions. Factors such as temperature, pH, and nutrient availability significantly influence the growth and metabolic activity of the microorganisms. Precise control of these parameters is crucial to maximizing biofuel production and minimizing the production of byproducts. A well-designed and controlled environment is essential to achieving high yield and efficiency in biofuel production.
Finally, the downstream processing of the biofuel is a key component of the overall production process. This step involves separating the biofuel from the microbial culture and further refining the product to meet the required specifications for its intended application. Efficient downstream processing is critical to minimizing losses and maximizing the economic viability of biofuel production.
Challenges and Future Directions
Despite the promising potential of microbial biofuel production, several challenges remain. One major hurdle is the need for cost-effective and scalable production methods. Developing efficient bioreactors and process optimization strategies is crucial for lowering the cost of biofuel production and making it economically competitive with traditional fuels.
Another challenge relates to the development of robust and efficient microbial strains for biofuel production. Genetic engineering and metabolic engineering approaches can be instrumental in enhancing the productivity and efficiency of the microorganisms. Further research into the genetic manipulation of microorganisms could lead to significant improvements in biofuel production.
Furthermore, integrating microbial biofuel production into existing infrastructure and supply chains requires careful consideration. The development of efficient and sustainable transportation and storage systems for biofuels is essential for widespread adoption. Addressing these challenges will be crucial for the successful commercialization of microbial biofuel technologies.
Optimizing Microbial Metabolism for Biofuel Synthesis
Harnessing Metabolic Pathways for Enhanced Biofuel Production
A crucial aspect of optimizing microbial metabolism for biofuel synthesis lies in understanding and manipulating the natural metabolic pathways within the microorganisms. This involves identifying key enzymes and regulatory mechanisms that govern the production of desired biofuel precursors. By strategically modifying these pathways, we can potentially enhance the efficiency of biofuel synthesis, leading to higher yields and reduced production costs. Researchers are exploring various genetic engineering techniques to achieve this, from introducing new genes encoding for specific enzymes to altering the expression levels of existing ones. This targeted approach aims to create microbes that are highly efficient in converting substrates into desired biofuels.
Furthermore, understanding the interplay between different metabolic pathways is essential. For instance, optimizing the carbon flow from sugar metabolism to biofuel biosynthesis is critical for maximizing the overall production efficiency. Careful consideration of the feedback mechanisms and regulatory networks within the cell is necessary to ensure that the desired metabolic pathways are prioritized, while unwanted pathways are suppressed. This comprehensive approach to metabolic engineering holds significant promise for boosting the performance of microbial biofuel factories.
Genetic Engineering Strategies for Microbial Optimization
Genetic engineering plays a pivotal role in optimizing microbial metabolism for biofuel synthesis. This approach allows scientists to precisely manipulate the genetic material of microorganisms, enabling them to produce biofuels more efficiently. Techniques like CRISPR-Cas9 gene editing provide powerful tools for targeted modifications, facilitating the introduction of genes encoding for improved enzymes or the modification of existing ones to enhance their catalytic activity. This allows for the creation of customized microbial strains tailored for specific biofuel production.
Beyond gene introduction, strategies focus on fine-tuning gene expression. Introducing promoters that enhance the expression of biofuel-related genes, or developing repressors to control the activity of competing pathways, can yield significant improvements in biofuel production. These strategies are crucial for achieving optimal biofuel yields and reducing the reliance on external factors that can influence microbial performance.
Metabolic Flux Analysis and Modeling for Biofuel Optimization
Understanding the intricate network of metabolic reactions within a microbe is crucial for optimizing its biofuel production. Metabolic flux analysis (MFA) is a powerful tool that allows us to quantify the flow of metabolites through different pathways. By analyzing these fluxes, researchers can identify bottlenecks in the metabolic pathways and pinpoint areas for improvement. This data-driven approach allows for a more targeted and efficient design of microbial strains that are optimized for biofuel production.
Computational modeling further enhances our understanding of microbial metabolism. Mathematical models can simulate the behavior of the microbe under different conditions, predicting the response to changes in substrate availability, environmental factors, and metabolic engineering interventions. These models can guide the design of experiments and help predict the outcomes of different genetic modifications, leading to more effective optimization strategies.
Substrate Utilization and Conversion Efficiency
Maximizing substrate utilization and conversion efficiency is paramount in optimizing microbial metabolism for biofuel synthesis. The ability of microbes to efficiently utilize various feedstocks like lignocellulosic biomass or agricultural residues directly impacts the overall cost-effectiveness of biofuel production. Strategies like metabolic engineering to enhance the uptake and degradation of these complex substrates are vital. This includes modifying the microbe's ability to break down complex polymers into simpler sugars that can be utilized for biofuel production.
Efficient conversion of these sugars into biofuel precursors is also key. Improving the enzyme activity and specificity involved in these conversion steps can significantly enhance the overall efficiency of the process. This requires a deep understanding of the kinetics and thermodynamics of the involved enzymatic reactions, enabling the development of targeted strategies to optimize the conversion process and increase the yield of the desired biofuels.
Environmental Factors and Strain Stability
Optimizing microbial metabolism for biofuel synthesis extends beyond genetic modifications and metabolic pathway engineering. Environmental factors, including temperature, pH, and oxygen concentration, significantly affect microbial performance. Understanding how these factors influence biofuel production is crucial. Optimizing these parameters can lead to increased yields and higher stability of the engineered microbial strains.
Maintaining the stability of engineered strains over time is essential for sustainable biofuel production. Developing strategies to prevent the reversion of engineered strains to their wild-type counterparts or the emergence of undesired mutations is critical. This includes identifying and implementing robust selection and maintenance strategies that ensure the consistent performance of the engineered microbes over multiple generations.
Developing Microbial Strains with Enhanced Performance
Optimizing Metabolic Pathways
A crucial aspect of developing microbial strains with enhanced performance lies in optimizing their metabolic pathways. This involves identifying and manipulating key enzymes and metabolic reactions to increase the production of desired products or enhance the efficiency of specific processes. By understanding the intricate network of metabolic reactions within a microbe, scientists can strategically target specific pathways for improvement, potentially leading to higher yields and faster production times. This targeted approach allows for more precise control over the metabolic output of the microbe, a significant advantage in industrial applications.
Further research into metabolic pathway engineering can uncover novel strategies to bypass bottlenecks in existing pathways or introduce entirely new pathways that enhance the production of valuable compounds. This could revolutionize the production of biofuels, pharmaceuticals, and other valuable chemicals.
Improving Genetic Stability
Maintaining genetic stability is paramount when engineering microbial strains. Introducing new genetic material into a microbe often leads to instability, where the desired traits are lost or the introduced genes are deactivated. Strategies to mitigate this instability are essential to ensure consistent and reliable performance of the engineered strain. Techniques like optimizing the integration sites for introduced genes and using strong promoters can greatly enhance the stability of the introduced genetic material. These strategies are crucial for long-term viability and effectiveness in industrial applications.
Robust genetic engineering approaches are needed to ensure the desired traits persist over successive generations. This requires a thorough understanding of the underlying mechanisms contributing to genetic instability in microorganisms.
Harnessing CRISPR-Cas Systems
CRISPR-Cas systems offer powerful tools for precise genetic manipulation, enabling researchers to modify microbial genomes with unprecedented accuracy and efficiency. These systems allow for targeted gene editing, gene deletion, and gene insertion, providing a precise and efficient method for tailoring microbial characteristics. The versatility of CRISPR-Cas systems makes them ideal for a wide range of applications in microbial strain engineering, ranging from modifying metabolic pathways to improving stress tolerance.
The ease of use and efficiency of CRISPR-Cas systems have significantly accelerated the development of engineered microbial strains. Researchers can now quickly and accurately introduce desired traits, leading to faster progress in the field of microbial engineering.
Enhancing Stress Tolerance
The ability of a microbial strain to withstand various environmental stresses, such as temperature fluctuations, nutrient limitations, and the presence of toxic compounds, is critical for its performance in industrial applications. Engineering microorganisms to enhance their stress tolerance can significantly expand their applicability in diverse environments and processes. Strategies for improving stress tolerance often involve modifying genes involved in stress response pathways, enhancing cellular protection mechanisms, or introducing genes from other organisms with superior stress tolerance.
Developing strains that thrive in challenging conditions is vital for successful implementation in bioreactors and other industrial settings. This resilience not only ensures the viability of the strain but also enhances the overall efficiency and productivity of bioprocesses.
Developing Robust Screening and Selection Methods
Efficient screening and selection methods are essential for identifying and isolating engineered microbial strains with improved performance. Developing robust and high-throughput screening methods is crucial for rapidly evaluating large numbers of potential strains and identifying those with the desired traits. These methods often involve innovative techniques like flow cytometry, high-content imaging, and automated screening platforms that can analyze numerous parameters simultaneously.
Implementing these advanced screening techniques can accelerate the entire strain development process, leading to the rapid identification of strains with superior performance characteristics. This, in turn, significantly reduces the time and resources required for strain optimization.
Utilizing Synthetic Biology Approaches
Synthetic biology principles play a crucial role in designing and constructing novel microbial strains with tailored functionalities. By combining genetic engineering techniques with computational modeling and systems biology approaches, scientists can create entirely new metabolic pathways and cellular functions. This approach allows for the design of microbes that are optimized for specific tasks, such as producing biofuels or degrading pollutants. Utilizing synthetic biology also enables the creation of microbial consortia with enhanced capabilities, further expanding the possibilities for industrial bioprocesses.
The ability to design and construct novel biological systems using synthetic biology principles has the potential to revolutionize a wide array of industries, enabling the production of sustainable and efficient bio-based products.
Proper hydration is crucial for numerous bodily functions, from regulating temperature to transporting nutrients. Maintaining adequate fluid intake is essential for overall health and well-being, impacting everything from cognitive function to physical performance. Dehydration can lead to fatigue, headaches, and impaired concentration, highlighting the vital role that liquids play in our daily lives.
Utilizing Synthetic Biology Approaches for Biofuel Production
Harnessing Microbial Diversity for Enhanced Biofuel Production
Synthetic biology offers a powerful toolkit to engineer microbes, allowing us to fine-tune their metabolic pathways to optimize biofuel production. This approach leverages the vast diversity of naturally occurring microorganisms, identifying and modifying specific genes or pathways for enhanced efficiency. By understanding the intricate mechanisms governing microbial metabolism, scientists can manipulate these processes to produce biofuels with higher yields and lower production costs, ultimately contributing to a more sustainable energy future.
Exploring different microbial species, from bacteria to algae, presents a wealth of possibilities. Researchers are investigating the potential of extremophiles, microorganisms thriving in harsh environments, for their unique metabolic capabilities. These organisms often possess enzymes and pathways capable of converting diverse feedstocks into biofuels, expanding the scope of sustainable energy production.
Optimizing Metabolic Pathways for Enhanced Biofuel Yield
A crucial aspect of synthetic biology in biofuel production is the optimization of metabolic pathways. Researchers can introduce or modify genes responsible for crucial steps in the biofuel production process, such as converting sugars into biofuel precursors. Precise manipulation of these pathways allows for the redirection of cellular resources towards biofuel synthesis, leading to increased yields and reduced byproducts.
This optimization process often involves creating or altering metabolic pathways to enhance the efficiency of substrate utilization. By strategically modifying enzyme activity and substrate specificity, scientists can improve the overall process, leading to higher biofuel titers and reduced costs associated with downstream processing.
Developing Robust and Efficient Biocatalysts
The development of robust and efficient biocatalysts is a critical component of synthetic biology approaches for biofuel production. Scientists are engineering microbes to produce enzymes with enhanced stability and activity under specific conditions, crucial for optimizing biofuel production at industrial scales. This involves modifying enzyme structures to improve their thermal tolerance, pH stability, and resistance to inhibitors, leading to increased production rates and reduced downstream processing complexities.
Utilizing CRISPR-Cas Systems for Targeted Gene Editing
CRISPR-Cas systems are revolutionizing genetic engineering, providing precise and efficient tools for modifying microbial genomes. This technology allows for targeted gene editing, enabling researchers to introduce specific mutations or add new genetic material to optimize biofuel production. Researchers can efficiently modify specific genes controlling metabolic pathways, improving substrate uptake, or enhancing the production of enzymes critical to biofuel synthesis.
Engineering Microbes for Tolerating Diverse Feedstocks
A significant challenge in biofuel production is the utilization of diverse and potentially low-cost feedstocks. Synthetic biology enables the engineering of microbes to effectively utilize a wider range of feedstocks. This involves tailoring microbial metabolism to efficiently break down lignocellulosic biomass, agricultural residues, or even waste materials into fermentable sugars, thereby expanding the range of available feedstocks for biofuel production.
Minimizing Environmental Impact of Biofuel Production
A crucial consideration in the development of biofuels is their environmental impact. Synthetic biology approaches can be used to reduce the environmental footprint of biofuel production. By engineering microbes to utilize waste materials as feedstocks, researchers can minimize the need for dedicated agricultural lands and reduce overall resource consumption. This approach focuses on minimizing the use of precious resources and reducing the environmental burden associated with biofuel production, making it a more sustainable alternative to fossil fuels.
Scaling Up Biofuel Production for Commercial Applications
The successful translation of synthetic biology approaches for biofuel production requires robust strategies for scaling up production to meet commercial demands. Developing efficient fermentation processes, optimizing downstream processing techniques, and ensuring cost-effectiveness are critical for widespread adoption. This involves creating bioreactors and fermentation strategies that can handle large-scale production while maintaining high yields and product quality. Further research and development are needed to make synthetic biology-based biofuel production economically viable and environmentally sound, making it a truly sustainable energy solution.
Challenges and Future Directions in Microbial Biofuel Engineering
Harnessing the Power of Microbial Diversity
Microbial communities, encompassing a vast array of species and metabolic capabilities, hold immense potential for biofuel production. Exploring and understanding the intricacies of these diverse microbial ecosystems is paramount. Researchers are actively investigating novel microbial consortia and individual strains with unique metabolic pathways, seeking to optimize their efficiency in converting various feedstocks into biofuels. This exploration extends beyond traditional model organisms, paving the way for harnessing the untapped potential of less studied microbial species.
Optimizing Metabolic Pathways for Enhanced Biofuel Production
Current microbial biofuel production processes often fall short of achieving maximum efficiency. Significant advancements are required in optimizing metabolic pathways within microorganisms to enhance the conversion of substrates into desired biofuel products. This involves manipulating gene expression, enzyme activity, and metabolic flux to direct the metabolic flow towards biofuel synthesis. Further research should focus on developing robust and scalable strategies for pathway engineering, ensuring sustained and efficient biofuel production.
Developing Robust and Scalable Bioreactor Designs
Scaling up biofuel production from laboratory settings to industrial applications requires innovative bioreactor designs. Current systems often face limitations in maintaining optimal growth conditions and product yields at larger scales. The development of efficient and cost-effective bioreactors, capable of handling high volumes of biomass and maintaining consistent process parameters, is crucial for the commercial viability of microbial biofuel production. This includes advancements in mixing, temperature control, and substrate delivery mechanisms.
Addressing Substrate Availability and Cost-Effectiveness
A major challenge in microbial biofuel engineering is ensuring a sustainable and cost-effective supply of feedstocks. Many current biofuel production methods rely on agricultural feedstocks, potentially competing with food production. Researchers are exploring alternative feedstocks, such as lignocellulosic biomass and industrial waste streams, to reduce reliance on food crops. Developing efficient methods for pre-treating these feedstocks to improve microbial accessibility and reduce processing costs is vital for wider application.
Improving Product Separation and Purification Techniques
Efficient separation and purification of biofuels from microbial fermentation broths are crucial for economic viability. Current methods can be energy-intensive and costly, hindering the widespread adoption of microbial biofuel technologies. Innovative separation and purification techniques, including membrane technologies, solvent extraction, and advanced chromatography, are needed to reduce processing costs and improve the purity of the final biofuel product. This will significantly impact the overall competitiveness of microbial biofuel production.
Overcoming Environmental and Economic Barriers
The environmental impact of microbial biofuel production needs careful consideration. Careful assessment of the entire life cycle, from feedstock sourcing to product utilization, is essential to minimize environmental footprint. Furthermore, the economic feasibility of microbial biofuel production must be assessed in comparison to traditional fossil fuel sources. Addressing factors such as land use, water consumption, and energy requirements is crucial for sustainable and economically viable biofuel production.
Enhancing Public Awareness and Acceptance
Public perception and acceptance of microbial biofuels are crucial for their successful integration into the energy sector. Educating the public about the environmental benefits, economic opportunities, and potential risks associated with microbial biofuel production is essential. Promoting transparency and accountability in biofuel production processes will foster public trust and support for this promising renewable energy technology. Open dialogue and engagement with stakeholders are essential for successful implementation.