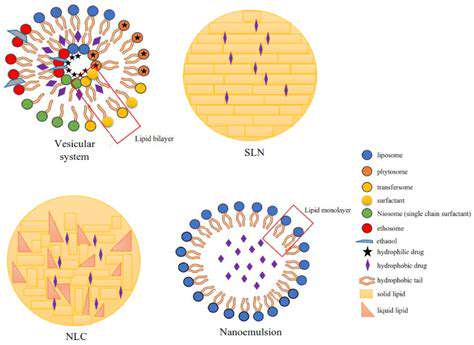
Nanocarrier Design and Synthesis
Nanotechnology offers a revolutionary approach to drug delivery, enabling the design of sophisticated nanocarriers that enhance drug efficacy and minimize side effects. These nanocarriers, ranging from liposomes to polymeric nanoparticles, are meticulously engineered to encapsulate therapeutic agents, targeting specific cells or tissues, and ultimately delivering the drug to the desired location within the body. The precise control over size, shape, and surface properties of these nanocarriers is crucial for optimal performance. This intricate design process involves careful selection of materials, synthesis techniques, and surface modifications to achieve the desired characteristics.
Various synthesis methods are employed to fabricate nanocarriers with the desired properties. These techniques include methods like emulsion polymerization, self-assembly, and nanoprecipitation. Each method presents unique advantages and challenges, influencing the final characteristics of the nanocarrier, such as its size distribution, drug loading capacity, and stability in biological environments. Careful control over the synthesis parameters is paramount to ensure consistent and reproducible nanocarrier production.
Targeted Drug Delivery
A key advantage of nanotechnology in drug delivery is the ability to target drugs to specific cells or tissues. This targeted delivery minimizes the systemic exposure of healthy tissues to the drug, reducing unwanted side effects. Nanocarriers can be engineered to selectively bind to specific receptors on target cells, ensuring that the drug is delivered precisely where it is needed. This precision targeting enhances therapeutic efficacy while minimizing toxicity.
This targeted delivery is achieved through various strategies. For instance, surface modifications of nanocarriers can be employed to incorporate targeting ligands, such as antibodies or peptides, that recognize specific receptors on diseased cells. This targeted approach allows for the preferential accumulation of the drug at the target site, maximizing treatment efficacy and minimizing side effects.
Drug Loading and Release Mechanisms
Effective drug loading is essential for the success of nanocarrier-based drug delivery systems. The ability to load a significant amount of drug into the nanocarrier while maintaining its stability and biocompatibility is critical. Different loading strategies, such as encapsulation, adsorption, and covalent conjugation, are employed to achieve optimal drug loading and release profiles. The controlled release of the drug from the nanocarrier is another crucial aspect of this technology, allowing for sustained and targeted delivery.
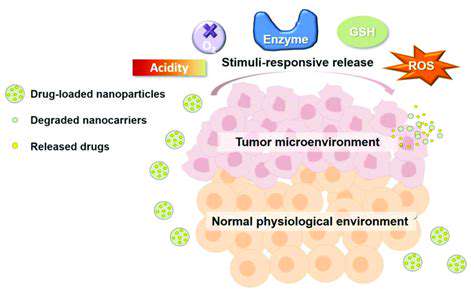
Different nanocarrier systems exhibit varying drug release mechanisms. For example, some systems release the drug in a burst, while others exhibit sustained release over a prolonged period. The choice of release mechanism depends on the specific therapeutic application and the desired drug concentration profile at the target site. The release mechanism is often dictated by factors such as the degradation of the nanocarrier or the stimuli-responsiveness of the material.
Clinical Applications and Challenges
Nanotechnology-driven drug carriers show great promise in various clinical applications, including cancer therapy, cardiovascular diseases, and infectious diseases. The ability to deliver drugs directly to diseased tissues, coupled with controlled drug release, significantly enhances therapeutic efficacy. Nanocarriers can overcome limitations of conventional drug delivery methods, such as poor drug solubility, rapid metabolism, and nonspecific targeting. This technology has the potential to revolutionize the treatment of numerous diseases.
Despite the promising potential, several challenges remain in translating nanocarrier-based drug delivery systems into clinical practice. These include ensuring the safety and biocompatibility of the nanocarriers, optimizing drug loading and release, and developing cost-effective manufacturing processes. Further research and development are needed to address these challenges and unlock the full therapeutic potential of nanotechnology-driven drug carriers.
Gene Therapy and Nucleic Acid Delivery: A Novel Approach
Gene Therapy: Revolutionizing Treatment Strategies
Gene therapy, a revolutionary approach in biotechnology, aims to treat genetic disorders by modifying or replacing faulty genes. This innovative field holds immense potential for curing a wide range of diseases, from inherited conditions like cystic fibrosis to acquired diseases such as cancer. By precisely targeting and delivering therapeutic genes, gene therapy offers a potential cure rather than just symptomatic treatment, promising a significant advancement in healthcare.
The fundamental principle behind gene therapy is to introduce functional genes into cells to compensate for the defective ones. This can be achieved through various methods, each with its own advantages and challenges. The success of gene therapy depends critically on the efficient delivery of these therapeutic genes to the target cells, highlighting the importance of effective nucleic acid delivery systems.
Nucleic Acid Delivery: A Critical Component
Nucleic acid delivery is a crucial step in gene therapy, as it determines the efficiency and safety of gene transfer. Successful delivery necessitates the development of vectors capable of transporting the therapeutic DNA or RNA to the target cells, while minimizing off-target effects and ensuring stability during transport.
Different types of vectors, including viral and non-viral vectors, are employed for nucleic acid delivery. Each type presents a unique set of advantages and disadvantages in terms of efficiency, safety, and cost. The choice of vector often depends on the specific application and the target cell type.
Viral Vectors: Nature's Delivery Systems
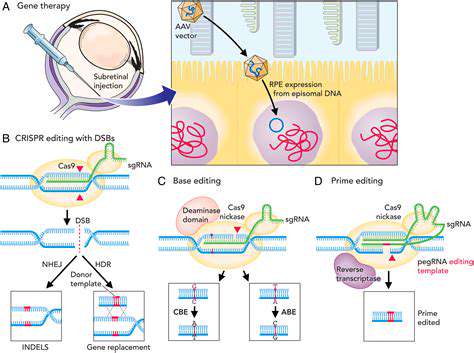
Viral vectors, derived from naturally occurring viruses, have emerged as powerful tools for gene delivery. Their natural ability to infect cells makes them potentially highly efficient at delivering therapeutic genes. However, viral vectors may trigger an immune response, potentially leading to rejection of the treatment or adverse effects.
Retroviruses, adenoviruses, and adeno-associated viruses (AAVs) are examples of viral vectors commonly used in gene therapy. Each type exhibits distinct tropism and characteristics, influencing their suitability for different applications. Further research is focused on improving the safety and targeting specificity of viral vectors.
Non-Viral Vectors: Safe and Efficient Alternatives
Non-viral vectors offer a promising alternative to viral vectors, often exhibiting a lower risk of immune responses. These vectors, such as liposomes and nanoparticles, encapsulate the therapeutic nucleic acids and deliver them to target cells. However, their efficiency in delivering genes may be lower compared to viral vectors, and further research is needed to improve their efficacy and target specificity.
Challenges and Future Directions
Despite the potential of gene therapy, several challenges remain. These include ensuring the safety and efficacy of gene delivery, addressing potential immune responses, and optimizing the long-term expression of introduced genes. Significant research efforts are underway to address these challenges, leading to the development of more refined and targeted therapies.
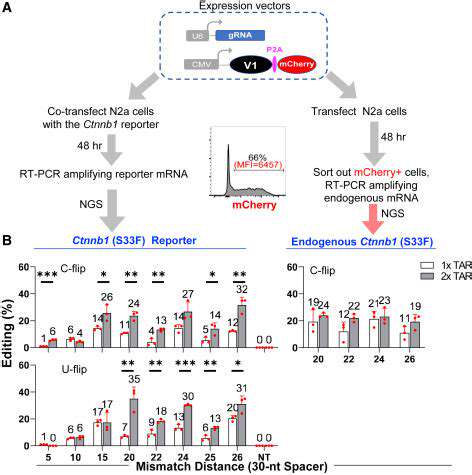
Advancements in gene editing technologies, such as CRISPR-Cas9, are further enhancing the precision and versatility of gene therapy approaches. These tools offer the potential to correct genetic defects with higher accuracy, opening new avenues for treating a broader spectrum of diseases.
Ethical Considerations in Gene Therapy
Gene therapy raises significant ethical considerations, particularly regarding the potential for germline modifications and the equitable access to these potentially life-altering treatments. Careful ethical review and robust regulatory frameworks are essential to ensure responsible development and application of gene therapy technologies.
The Role of Biotechnology in Nucleic Acid Delivery
Biotechnology plays a crucial role in developing novel nucleic acid delivery systems. Researchers are constantly exploring innovative approaches to enhance the efficiency, safety, and targeting capabilities of vectors. These advancements are essential to unlock the full therapeutic potential of gene therapy and pave the way for future breakthroughs in healthcare.
Space-based observatories represent a paradigm shift in astronomical observation, offering unparalleled opportunities to study celestial phenomena free from the limitations of Earth's atmosphere. These orbital platforms, equipped with sophisticated instruments, provide clearer and more detailed images of distant galaxies, nebulae, and other cosmic structures. The absence of atmospheric distortion allows for precise measurements and observations, enabling astronomers to probe the fundamental workings of the universe with a level of accuracy previously unattainable. The ability to observe in wavelengths blocked by Earth's atmosphere, such as infrared and ultraviolet light, reveals a wealth of information about celestial objects that would otherwise remain hidden.
3D Bioprinting for Customized Drug Delivery Systems
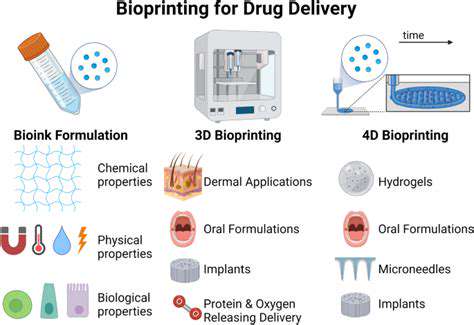
3D Bioprinting: A Revolutionary Approach
3D bioprinting is rapidly emerging as a transformative technology with the potential to revolutionize various fields, including drug discovery and personalized medicine. It allows for the creation of complex, three-dimensional structures mimicking biological tissues, opening exciting avenues for customized drug testing and development. This precision approach offers a significant advancement over traditional methods, promising faster and more efficient drug development cycles.
The ability to precisely control the arrangement and composition of biomaterials within these 3D constructs is a key advantage. This allows researchers to create highly controlled environments that closely mimic the intricate architecture of human tissues, thereby providing a more accurate representation of how drugs interact with biological systems. This is a critical advancement, especially in the context of personalized medicine, where tailored drug responses are paramount.
Customized Drug Delivery Systems
One of the most significant applications of 3D bioprinting in drug development lies in creating customized drug delivery systems. These systems can be designed to release drugs at specific rates and locations within the body, leading to improved therapeutic efficacy and reduced side effects. This precise control over drug release is crucial for maximizing the effectiveness of therapies and minimizing harmful interactions with healthy tissues.
By incorporating drug-loaded bioinks into the 3D printed constructs, researchers can create sophisticated delivery systems that target specific cells or tissues. This targeted delivery approach is particularly important for treating diseases affecting specific organs or regions of the body.
Personalized Drug Testing
3D bioprinting enables the creation of personalized drug testing models, allowing researchers to evaluate the efficacy and safety of drugs in a more realistic setting. These models can be tailored to individual patient characteristics, including genetic predispositions and disease-specific features. This approach is crucial for predicting drug response and identifying potential adverse effects in a personalized manner.
This personalized approach to drug testing is a major step forward in pharmaceutical research, as it allows for more accurate predictions of drug efficacy and side effects. It represents a significant advancement over traditional methods, which often fail to capture the complexity of individual patient responses.
Accelerated Drug Development
3D bioprinting holds the potential to drastically accelerate the drug development process. By enabling the rapid creation of complex tissue models, researchers can significantly reduce the time and resources required to evaluate drug candidates. This accelerated process is critical for bringing new therapies to patients more quickly, especially for conditions with unmet medical needs.
The ability to rapidly iterate and refine drug designs based on real-time data from bioprinted models is also crucial for optimizing drug efficacy and safety profiles.
Improved Understanding of Drug Mechanisms
3D bioprinting can facilitate a deeper understanding of drug mechanisms of action. By visualizing how drugs interact with cells and tissues in a controlled environment, researchers can gain valuable insights into the molecular processes involved. This enhanced understanding can lead to the development of more effective and targeted therapies.
By creating highly controlled microenvironments mimicking the human body, 3D bioprinting allows for a detailed analysis of drug-tissue interactions, providing valuable insights into drug behaviour and efficacy.
Ethical Considerations and Future Directions
While 3D bioprinting presents significant opportunities for drug development, ethical considerations must be addressed. Ensuring the safety and efficacy of bioprinted drug delivery systems and personalized drug testing models is crucial. Furthermore, issues of accessibility and cost-effectiveness need to be carefully evaluated.
Future research should focus on developing standardized protocols and validation methods for 3D bioprinted models. This will ensure the reliability and reproducibility of results, facilitating the widespread adoption of this innovative technology in drug development.
Biodegradable Polymers and Bioconjugation Strategies: Enhancing Stability and Targeting
Biodegradable Polymer Selection for Enhanced Stability
The selection of appropriate Biodegradable Polymers is crucial for achieving desired stability characteristics in bioconjugates. Different polymers offer varying degradation rates, mechanical properties, and biocompatibility profiles. Polylactic acid (PLA), polycaprolactone (PCL), and poly(ethylene glycol) (PEG) are frequently used due to their biocompatibility and tunable degradation rates. Careful consideration of the specific application, including the intended degradation timeframe and the environment in which the bioconjugate will reside, is vital for choosing the most suitable polymer backbone.
Factors such as the polymer's molecular weight, crystallinity, and the presence of functional groups significantly influence its degradation kinetics and stability within the biological system. Researchers often employ techniques like enzymatic degradation studies and in vitro degradation assays to evaluate the performance of various polymer candidates under simulated physiological conditions. This meticulous selection process ensures the bioconjugate maintains its structural integrity and functionality throughout its intended lifespan.
Strategies for Bioconjugate Synthesis
Effective bioconjugation strategies are essential for linking the therapeutic payload or targeting moiety to the biodegradable polymer. Different approaches, such as covalent bonding, non-covalent interactions, and supramolecular assemblies, can be employed depending on the desired properties of the final bioconjugate. Careful consideration of the chemical reactivity of the polymer and the targeting ligand is paramount to ensure successful conjugation without compromising the stability or functionality of either component.
Chemical modification of the polymer backbone to introduce functional groups compatible with the targeting molecule is often necessary. Specific coupling reactions, such as click chemistry or carbodiimide chemistry, provide controlled and efficient methods for linking the components, thereby enhancing the stability and preventing unwanted side reactions. Optimizing these synthetic protocols is crucial for achieving high yields and minimizing potential side products.
Targeting Moieties for Enhanced Biodistribution
The incorporation of specific targeting moieties is a critical aspect of bioconjugate design, enabling the selective delivery of therapeutic agents to diseased tissues or cells. Antibodies, peptides, or aptamers can be conjugated to the polymer to achieve targeted delivery. These targeting molecules recognize specific receptors or antigens expressed on the surface of target cells, facilitating the accumulation of the bioconjugate at the desired site of action. This targeted delivery minimizes off-target effects, enhancing the therapeutic efficacy and safety of the drug.
Evaluation of Bioconjugate Stability in Biological Environments
Assessing the stability of bioconjugates in complex biological environments is paramount to ensure their efficacy and safety. In vitro studies using cell cultures and in vivo animal models are essential tools to evaluate the bioconjugates' behavior in the presence of enzymes, proteins, and other biological components. These studies allow for the identification of potential degradation pathways, the estimation of the half-life of the bioconjugate, and the evaluation of the release kinetics of the therapeutic payload.
Analyzing the stability of bioconjugates under various physiological conditions, including different pH values, temperatures, and the presence of reducing agents, is necessary. These investigations help to optimize the bioconjugate's performance and ensure its ability to maintain its integrity and function throughout its journey within the biological system. This comprehensive evaluation ensures that the bioconjugate remains stable and effective, delivering its payload as intended.
Optimizing Bioconjugate Properties for Enhanced Therapeutic Efficacy
Optimizing the properties of biodegradable polymers and bioconjugates is crucial for enhancing their therapeutic efficacy. Factors such as the degradation rate, the controlled release of the therapeutic agent, and the targeting specificity need to be carefully considered. Modifying the polymer's structure, incorporating stimuli-responsive elements, and fine-tuning the targeting moieties can significantly enhance the therapeutic efficacy of the bioconjugate. These modifications can lead to improved drug delivery, reduced side effects, and enhanced treatment outcomes.
Furthermore, the development of advanced characterization techniques allows for a deeper understanding of the bioconjugate's behavior in the biological system. These techniques enable monitoring of the bioconjugate's degradation, localization, and release profile, providing valuable insights for optimizing design parameters. This thorough optimization process leads to improved efficacy, reduced toxicity, and ultimately, better treatment outcomes for patients.